

The high-efficiency filters (HEPA/ULPA) for the air supply ceiling are made of ultra-fine fiber structure. They must not be washed, wiped forcefully, or blown with high pressure. Otherwise, the pore structure of the filter material will be damaged, resulting in filter material damage and leakage, and the filtering efficiency will be directly lost. The “surface cleaning” only targets the frame, outer mesh, and sealing rubber ring of the filter. The specific operation process is as follows: 1. Preparations before cleaning Stop the machine and cut off the power supply: Close the air supply ceiling fan and the corresponding power supply, wait until the fan completely stops before operating. Avoid air flow carrying dust particles into the deep layer of the filter material. Personnel protection: Operators must wear clean clothes, dust-free gloves, and protective masks to avoid skin oils and hair contamination of the filter and the clean room environment. Tool preparation: Prepare non-fibrous dust-free cloth, purified water (or clean room-specific neutral cleaning agent), and dust-free wiping rods. Do not use ordinary rags, detergent, alcohol, etc., which are corrosive or prone to shedding. 2. Cleaning process by parts Filter frame cleaning Dip a small amount of purified water into the dust-free
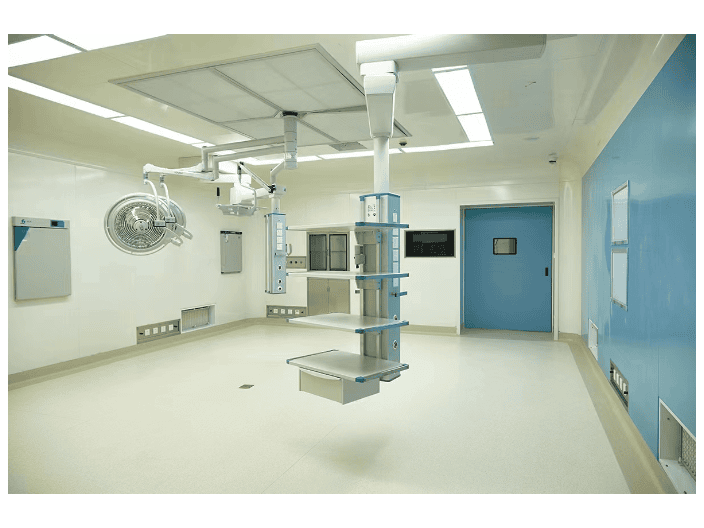
The record management for the daily maintenance of the air supply ceiling is a crucial part for ensuring the stable operation of the equipment, tracing the root causes of problems, and optimizing maintenance strategies. The recorded content should be comprehensive, accurate, and traceable, and it should be linked with the overall operation archives of the cleanroom. It specifically includes the following six modules: Equipment basic information module Core parameters of the air supply ceiling: equipment model, factory serial number, installation location (such as XX workshop XX area of the cleanroom), rated air volume, matching filter specifications (initial efficiency / medium efficiency / high efficiency models, filtration efficiency, initial resistance). Maintenance ledger number: unique ledger code compiled by year / area for easy archiving and retrieval. Daily maintenance execution record module Maintenance time: precise to year / month / day / hour, distinguishing daily inspections, weekly deep maintenance, monthly specialized maintenance, and quarterly sealingity tests. Maintenance personnel: operator’s name, position, with signature confirmation, clearly defining the responsible party. Maintenance content and results: Daily inspection: fan operation status (no abnormal noise, vibration, stable rotation speed), diffuser plate sealing condition, surface cleaning areas and used tools (such as dust-free cloth + purification water).

A clean bench is a basic equipment that provides a localized sterile and dust-free clean environment. Through the use of a fan to filter the air through an efficient filter, a unidirectional airflow is formed, isolating external contaminants and being widely used in various industries with high requirements for environmental cleanliness. The core of daily maintenance lies in ensuring the cleanliness effect and prolonging the equipment’s service life. 1. Core application fields Biomedical and life science field This is the main application scenario of the clean bench, used for sterile operation-related experiments and production. For example, cell culture, bacterial inoculation and purification, genetic engineering experiments, sterile formulation assembly, vaccine preparation, etc., it can effectively prevent samples from being contaminated by microorganisms and protect operators from harmful bacterial strains. Microbial detection and food industry In the sterile detection laboratories of food enterprises, it can be used for the isolation and cultivation of total bacterial count and pathogenic bacteria in food; it is also suitable for the preparation of probiotic preparations and sterile food ingredients, ensuring that the microbial indicators of the products meet the standards. Electronic and semiconductor industry It is used for the assembly, inspection and maintenance of precision electronic

The biopharmaceutical industry has extremely strict requirements for aseptic environments and prevention of cross-contamination. The ultra-clean workbench, as a key equipment for core processes such as sterile formulation production, bacterial strain operation, and cell culture, must strictly follow standardized operating procedures to ensure product quality complies with GMP (Good Manufacturing Practice for Pharmaceuticals) standards. This specification covers four parts: pre-operation preparation, operation control during the process, post-operation processing, and special scenarios. I. Pre-operation Preparation Personnel Preparation Strictly follow the changing procedures for the clean area in biopharmaceuticals: change shoes → remove outer clothing → put on clean clothing → wear a mask and gloves → hand disinfection (75% ethanol wiping, action ≥ 30 seconds). It is strictly prohibited to wear jewelry, watches, and long hair must be completely contained in a clean cap to avoid hair and skin debris from falling into the workbench. Avoid strenuous exercise, eating, and makeup 30 minutes before entering the clean area to reduce microbial emissions. Equipment Inspection and Self-Cleaning Appearance and condition: Check the workbench surface, inner wall, and air outlet grille for cleanliness, without residual stains or debris; check the equipment power supply, control panel for normal operation, and no abnormal alarm on

The service life of the embedded clean transfer window is not fixed and mainly depends on the material quality, usage frequency, maintenance level, and the cleanliness level of the environment. The industry-wide reference range is as follows: Main structure service life Stainless steel (304/316 stainless steel): Strong corrosion resistance and deformation resistance. Under normal maintenance, the main frame can last for 8-12 years. In some cases (low frequency use + regular maintenance), it can be extended to 15 years. Carbon steel spray-painted material: Weak corrosion resistance, prone to paint peeling and rusting. The service life is usually 5-8 years. Life of key components (need to be replaced regularly) The core functional components of the embedded clean transfer window are consumables or wearables, and their lifespan is much shorter than that of the main structure. Regular replacement is necessary to ensure the cleanliness performance: High-efficiency filter: Core consumable of the clean room. Depending on the usage environment and pressure difference changes, the replacement cycle is 3-6 months to 1 year. Sealing strip: Used to ensure the sealing performance of the transfer window. Affected by environmental temperature and humidity and friction, the aging period is 1-2 years. Interlock device (mechanical/electronic): Mechanical interlock

To determine whether the service life of the clean transfer window has expired, it is not sufficient to only consider the duration of use. A comprehensive judgment should be made by taking into account the state of the main structure, the performance of core components, and the compliance of the clean function. The specific judgment criteria are as follows: Judgment of aging / damage of the main structure Material corrosion and deformation: Large areas of pitting on the stainless steel frame, weld cracks, or peeling of the paint on the carbon steel spray-painted shell, and penetration of rust through the sheet; cavities show depressions and deformations, which cannot ensure a sealed space, indicating that the structure has expired. Abnormal operation of doors and windows: Doors and windows are stuck, unable to fully close, or the gap between the door frame and the cavity exceeds 0.5mm, and even after replacing the sealing strips, the problem cannot be improved, requiring a complete replacement. Failure of core components Interlock device failure: Mechanical interlock cannot achieve the function of “one door opening, the other door locked”; electronic interlock frequently malfunctions, sensors have no response, and the fault recurs after multiple repairs. Performance attenuation of

The core differences between the ordinary high-efficiency supply air outlet and the DOP liquid tank high-efficiency supply air outlet lie in the sealing structure, leak detection method and applicable cleanliness level. The detailed differences between the two can be compared from the following five dimensions: Comparison dimension Ordinary high-efficiency air supply outlet DOP liquid tank high-efficiency supply air outlet Sealing method It adopts a compression-type seal. Through sponge and rubber sealing strips, the filter frame is compressed by the frame bolts to achieve sealing. This is a “dry seal”. The liquid tank seal is adopted. The frame is equipped with a dedicated liquid tank adhesive. The filter frame is inserted into the liquid tank adhesive to form a liquid seal, which belongs to “wet-type sealing”. Leak detection method The conventional dust particle counter is used for scanning and leak detection, to measure the particle leakage at the connection point between the filter and the frame. Specialized DOP/PAO dust leakage detection. By using an aerosol generator to release particles, the leakage rate at the sealed area can be detected, with higher accuracy. Sealing performance The sealing performance is moderate. Due to the aging of the sealing strips and uneven compression force,

There is no fixed standard for the replacement cycle of the liquid tank sealant of the high-efficiency air outlet. The core factors depend on the quality of the sealant, the usage environment, and the test results of the sealing performance. The industry’s general reference and judgment basis are as follows: Regular replacement cycle High-quality liquid tank sealant (such as polyurethane-based liquid tank sealant with aging resistance and anti-shrinkage properties) can be replaced once every 2-3 years in a stable environment of a 100-level or higher cleanroom; If using ordinary liquid tank sealant or if the cleanroom has large temperature fluctuations, corrosive gases, or high dust concentration, it is recommended to replace it once every 1-2 years. Criteria for immediate replacement Visual inspection: If the liquid tank sealant shows hardening, cracking, shrinking, flowing, or there are gaps at the bonding point with the frame; Leak detection test: When using DOP/PAO leak detection, if the leakage rate at the filter and liquid tank sealing area exceeds the standard, and the filter itself is not damaged, the liquid tank sealant needs to be replaced; Maintenance operation: When replacing the high-efficiency filter, it is recommended to check the condition of the liquid tank sealant

The air purification and disinfection machine is a device that integrates air purification and sterilization and disinfection. It can remove particles, odors, harmful gases, and kill bacteria, viruses and other microorganisms at the same time. It is widely used in various scenarios. Its daily maintenance directly affects the operational efficiency and service life of the equipment. I. Core Application Fields 1. Medical and health care field Applicable scenarios: Hospital outpatient departments, wards, operating rooms, laboratories, disease control center laboratories, etc. Core requirements: The hospital environment is densely populated with pathogenic microorganisms. The equipment must have highly efficient sterilization capabilities (such as killing the novel coronavirus, influenza virus, Staphylococcus aureus, etc.), and at the same time filter out surgical smoke and odors from drugs to meet the hospital’s infection control standards. Special requirements: In some scenarios, models that comply with the “Regulations on Hygiene and Safety Evaluation of Disinfection Products” should be selected to support coexistence with humans and avoid excessive ozone. 2. Civilian home and office fields Applicable scenarios: Home interiors, office buildings, school classrooms/dormitories, kindergartens, nursing homes. Core requirements: Remove formaldehyde, PM2.5, pollen, and pet hair, kill mites and mold spores, and relieve discomfort for people with allergies. In

To determine whether the filter of an air purification and disinfection machine needs to be replaced, four dimensions should be considered: filter type, usage duration, equipment performance, and sensory experience. The criteria for judging different filters vary significantly. The specific methods are as follows: 1. Make targeted judgments based on the type of filter screen 1. Primary filter screen (washable, needs to be replaced regularly The primary filter screen mainly intercepts hair and large particles of dust. The core basis for determining replacement is the filtration efficiency after cleaning and the condition of the filter screen If the filter screen is still deformed, damaged or has pilling fibers after cleaning, and cannot closely fit the card slot, unfiltered air will directly enter the interior of the equipment. At this point, it needs to be replaced. Within the same period, if the dust accumulation speed at the air inlet significantly accelerates, or if the air volume decreases within 1-2 weeks after cleaning, it indicates that the filter mesh diameter has been clogged and lost its filtering capacity. Regular replacement cycle: After repeated cleaning 3 to 5 times, it is recommended to replace even if the appearance is intact. In highly polluted

The supply air ceiling of the operating room is the core terminal equipment of the air conditioning system in the clean operating room. It achieves a high cleanliness and low bacterial concentration environment in the operating area through reasonable air flow organization and is a key facility to ensure surgical safety. Its application and maintenance must strictly follow the relevant norms of the clean operating department (such as GB 50333-2013 “Technical Code for Hospital Clean Operating Department Buildings”). I. Core Applications of Air Supply Ceilings in Operating Rooms 1. Application scenarios and compatibility levels Supply air ceilings are mainly used in Class I to III clean operating rooms. Different grades of operating rooms have different performance requirements for them Class I (Class 100) operating room: It is suitable for surgeries with extremely high aseptic requirements such as cardiac surgery and neurosurgery. It should adopt a vertical laminar flow type air supply ceiling. The airflow should flow vertically downward at a speed of 0.25 to 0.30m/s, evenly covering the operating table to form a “clean air flow barrier” and isolate surrounding contamination. Class II to III (Class 1,000 to 10,000) operating rooms: Suitable for routine surgeries in orthopedics, general surgery, etc.
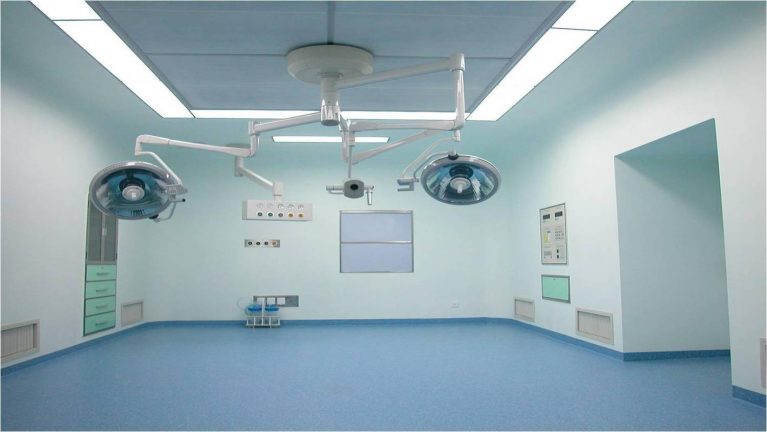
To determine whether the high-efficiency filters (HEPA/ULPA) of the supply air ceiling in the operating room need to be replaced, it is necessary to combine three core dimensions: differential pressure monitoring, performance testing, and service life, and at the same time follow the requirements of GB 50333-2013 “Technical Code for Building Clean Operating Rooms in Hospitals”. The specific judgment method is as follows: 1. Differential pressure monitoring (core judgment basis) The resistance of high-efficiency filters will increase with the increase of dust accumulation. The pressure difference is the most direct indicator for judgment. The operation steps are as follows: Record the initial pressure difference After the new filter is installed and the self-cleaning operation meets the standards, record the initial pressure difference before and after the high-efficiency filter. This value will be retained as a reference value. Daily monitoring comparison During normal operation, regularly check the reading of the differential pressure gauge that comes with the supply air ceiling. When the actual pressure difference reaches 1.8 to 2 times the initial pressure difference, replacement should be arranged immediately. If the pressure difference exceeds twice the initial pressure difference and the operation continues, it will cause a sudden drop in the

If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as wecan.